Umxholo
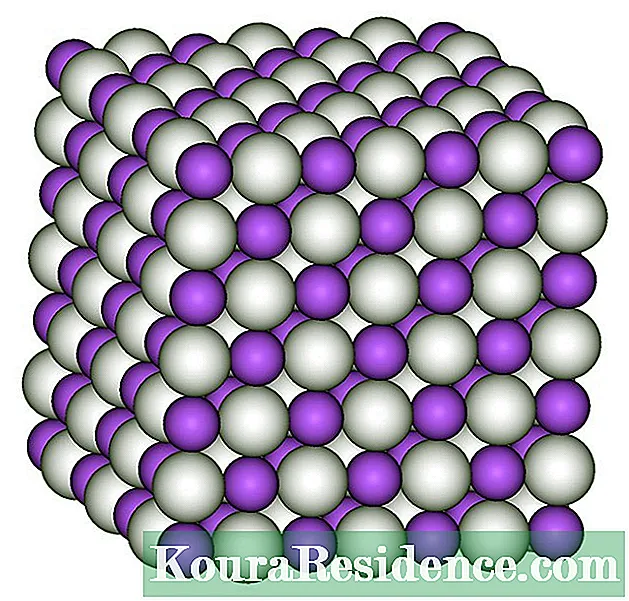
Inkqubo ye- amakhonkco esinyithi Luhlobo lomanyano lweekhemikhali olwenzeka phakathi kweeathom zentsimbi enye, kwaye apho kufezekiswa khona izakhiwo zeemolekyuli ezixineneyo, kuba i-nuclei ye iiathom badibana kakhulu kangangokuba baqala ukwabelana ngee-elektron zabo ze-valence.
Ezi zokugqibela zishiya imijikelo yazo yesiqhelo kwaye zihlala zijikeleze iseti ye-nuclei, njengohlobo lwelifu, kunye nomtsalane phakathi kweentlawulo zabo ezimbi kunye neentlawulo ezilungileyo ze-nuclei yiyo ebambe iseti imanyene.
Ngele ndlela, Ubophelelo lwentsimbi luxinene kwaye luyimbophelelo yeatom, ezinokuthi zenzeke kuphela phakathi kwee-atom zohlobo olunye kwaye zingaze zibe luhlobo lwealloy. Kananjalo ezi ntlobo zekhonkco akufuneki zibhidaniswe nazo ionic okanye covalent, nangona le yokugqibela inokwabelana ngemiba ethile kuba iiathom zazibandakanya ukutshintshiselana kwee-electron ukuya kwinqanaba elithile.
Iipropati zebhondi zetsimbi
Kwimeko yeebhendi zentsimbi uninzi lweempawu zesinyithi zibangelwa, njengokuqina kunye nobulukhuni bezinto zabo, ukuthamba kwayo kunye ductility, ulungile ukuqhuba kobushushu okanye umbane, kunye nokubengezela kwabo, kuba babuyela phantse kuwo onke amandla okukhanya ababethayo.
Amasuntswana eathom ahlangeneyo ngolu hlobo lwemixholo ahlala ecwangciswe macala omathathu kwi-hexagonal, cubic structures, okanye ngezinye iindlela ezininzi: njengakwimeko ye imekyuriUmzekelo, ulwelo kubushushu bamagumbi, umanyano lweathom lwenzeka ngeendlela ezahlukeneyo kwaye luvumela ukuyilwa kwamaconsi ajikelezileyo ale ntsimbi.
Imizekelo yeebhondi zentsimbi
Iibhondi ze-Metallic zihlala zihlala kwihlabathi le-athomu lezinyithi, ngoko ke nayiphi na into ecocekileyo yensimbi ngumzekelo onokuthi, umzekelo:
- Amakhonkco phakathi kwee-athomu zesilivere (Ag).
- Amakhonkco phakathi kwee-athomu zeGolide (Au).
- Amakhonkco phakathi kwe-Cadmium (Cd) athom.
- Amakhonkco phakathi kwee-atom ze-Iron (Fe).
- Iibhondi phakathi kweeatom zeNickel (Ni).
- Iibhondi phakathi kweeZinc (Zn) iiathom.
- Iibhondi phakathi kweeathom zeCopper (Cu).
- Iibhondi phakathi kweePlatinam (Pt) iiathom.
- Amakhonkco phakathi kweeathom zeAluminiyam (Al).
- Unxibelelwano phakathi kweeathom zeGallium (Ga).
- Iibhondi phakathi kweeathom zeTitanium (Ti).
- Iibhondi phakathi kweeathom zePalladium (Pd).
- Unxibelelwano phakathi kwee-atom ze-Lead (Pb).
- Iibhondi phakathi kweeathom zeIridium (Ir).
- Iibhondi phakathi kwee-atob zeCobalt (Co).
Unokukukhonza
- Imizekelo yeebhondi ze-Ionic
- Imizekelo yeebond Bond